The UK’s bizarre $1.7 billion vaccine rug pull

Back in February, biotech firm Valneva SE’s proposed COVID-19 vaccine was touted by Boris Johnson’s government as a key plank of its ambitious, whatever-it-takes race to immunize the Brits.
The U.K. had poured millions of pounds into Valneva’s Scottish factory, secured an extra 40 million vaccine doses on top of the 60 million it had already agreed to buy, and boasted about the vaccine’s potential as a booster in the autumn. The fact that Valneva was headquartered in France, whose vaccine rollout was off to a dire start and where zero doses had been procured, was the ultimate punchline.
Now the U.K. seems to have shot itself in the foot, rather than in the arm.
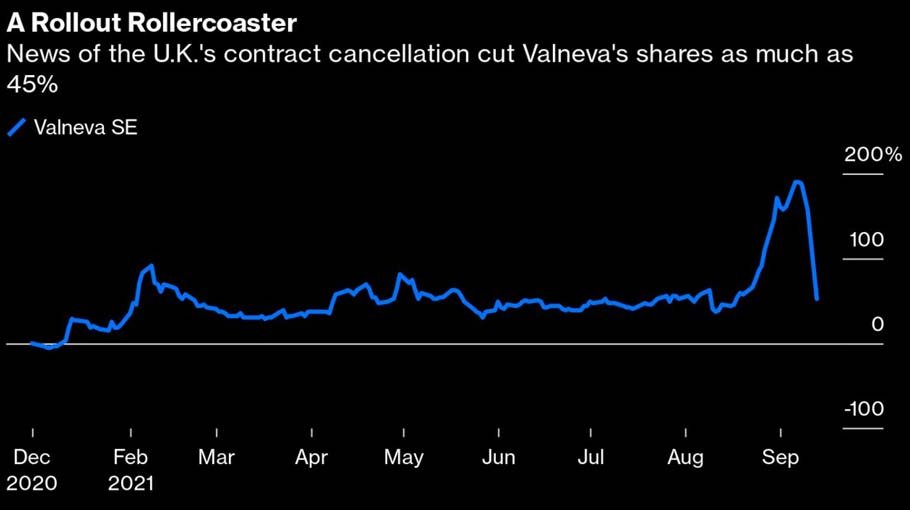
The government is abruptly canceling its supply contract, having found Valneva in breach of its obligations without specifying how — something Valneva is contesting, though without giving more details. Valneva’s stock, having almost tripled this year on the back of the U.K.’s endorsement, fell as much as 45% in Paris trading on Monday, wiping roughly $1 billion off its market value. The contract’s potential worth of up to €1.4 billion ($1.7 billion) accounted for almost half of Valneva’s estimated fair value, according to RX Securities analyst Samir Devani.
It’s hard to understand the reason for the rug-pull. One might at least assume that if there was a serious problem with the vaccine, Valneva would have to disclose it — but there’s been no such announcement. A minor delivery delay is one possibility that would not require additional disclosure, according to Kempen analyst Ingrid Gafanhao, and would be consistent with the company’s reference to having worked “to its best efforts.” Valneva declined to comment further.
The U.K., meanwhile, is describing this as an “ongoing commercial issue” and shrugging it off as having no impact on its vaccine rollout plans this year. As cloudy as this situation is, that’s a big change in tone.
It’s hard to dismiss the possibility that a change of political winds — from “whatever it takes” to “we’re alright, thanks” — is what’s behind the decision. Britain has several hundred million doses ordered from other manufacturers including Pfizer Inc.-BioNTech SE, which is already signing agreements on booster shots.
Even though Valneva’s Phase III clinical trial results haven’t been published yet, the hurdle is high for new vaccines in a market where efficacy rates of tried-and-tested shots are strong and severe cases continue falling. Governments may simply be less willing to put up cash for more risky vaccine ventures.
If that’s the case, this looks like a pretty clumsy milestone in a rollout that the U.K. has trumpeted as a post-Brexit success story. The country has trashed its own investment, shaken confidence in a new generation of vaccines — the French head of AstraZeneca Plc called it a “blow” — and snubbed a chance for a pragmatic renegotiation. The European Union has had its own vaccine bust-ups with AstraZeneca, but it resulted in a delivery settlement and a stream of supply for the Covax initiative to vaccinate the developing world. Maybe the U.K. could have made a similar move here.
Consider also that as recently as April, two months after exercising an option to purchase more doses, then-U.K. Health Secretary Matt Hancock praised the results of Valneva’s early vaccine trials and its potential to “play an important role” in protecting people if approved by regulators. What’s changed since then, aside from the pandemic curve and an improved supply of vaccines from elsewhere, isn’t yet clear.
There’s a lot we still don’t know about this story, but it’s a sign that the political and financial capital being spent on COVID-19 vaccine bazookas isn’t limitless.
In a pandemic where swathes of the world lack access to vaccines while others gear up for boosters, we should hope that the shift from confidence to caution doesn’t turn into complacency.
Lionel Laurent is a Bloomberg Opinion columnist covering the European Union and France.
Source: Bloomberg